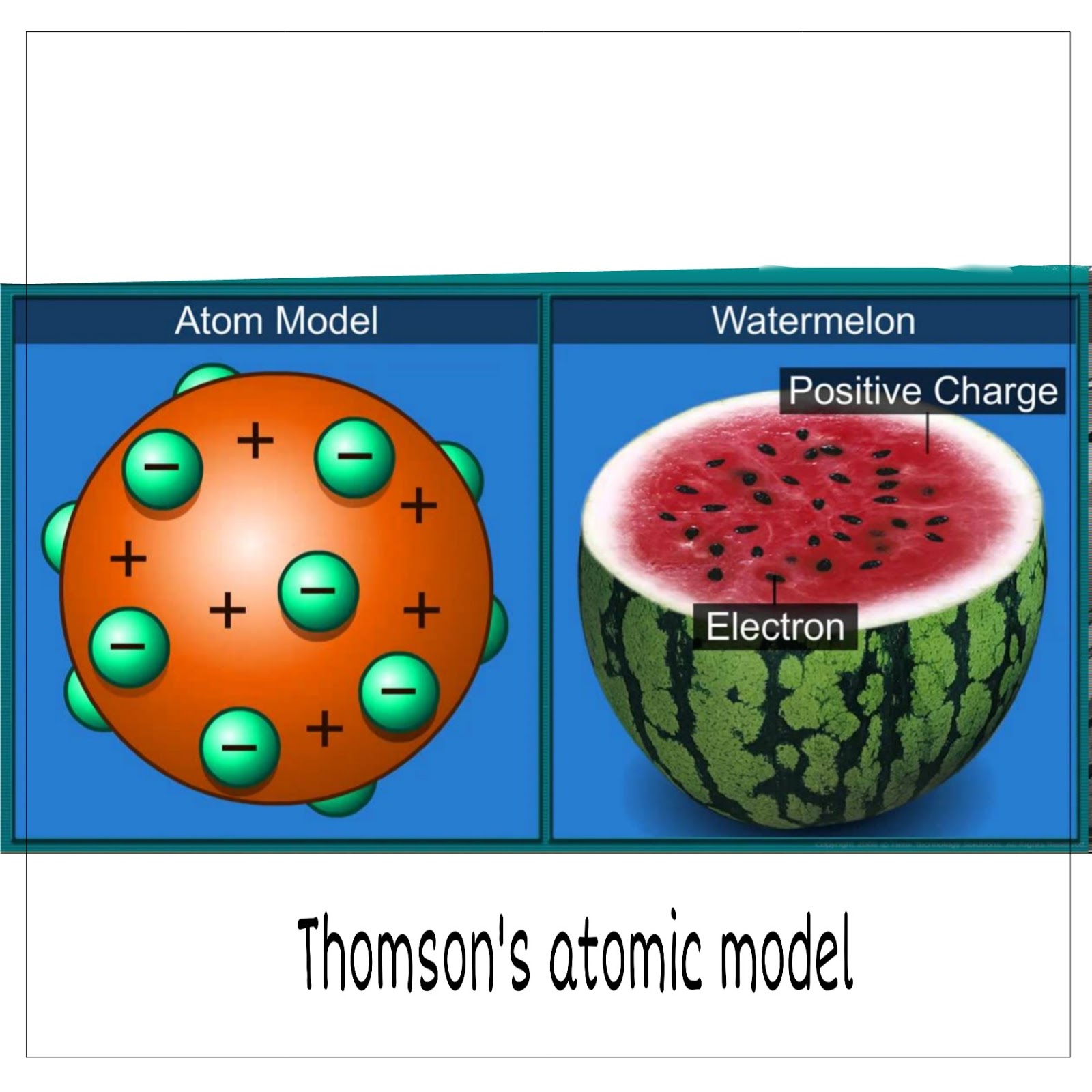
Structure of Atom: The discovery of two basic particles (electrons and protons) inside the atom caused Dalton’s atomic theory to collapse. Thomson was the first to suggest an atomic structure model. Many scientists developed numerous atomic theories to explain this. It was thought vital to understand how electrons and protons are ordered within an atom. Atomic Structure: The finding of two basic particles (electrons and protons) inside the atom caused Dalton’s atomic theory to collapse. As a result, several scientists presented different atomic models. The question of how these particles are organized within an atom arises. However, the atom’s discovery of electrons and protons caused this idea to collapse. The plum pudding atomic theory assumed that an atom’s mass is equally distributed across the atom.Īccording to Dalton’s atomic hypothesis, the atom is indivisible and indestructible. The implanted electrons resembled the seed of a watermelon, and the red bulk of the watermelon reflected the positive charge distribution. Thomson’s atomic model was sometimes referred to as the plum pudding or watermelon model. The positively charged particles are dispersed equally, while the electrons are placed in such a way that the atom is electrostatically stable. Thomson claimed that the form of an atom is similar to that of a sphere with a radius of 10-10 m. Thomson was successful in describing an atom’s total neutrality. In his atomic structure model, atoms were surrounded by a cloud with both positive and negative charges. He thought an atom was made up of thousands of electrons and that an electron was two thousand times lighter than a proton. We may also conceive of this as a watermelon, with the positive charge in the atom scattered all over like the red edible portion of the watermelon, and the electrons lodged in the positively charged sphere like the seeds. The electrons in a positively charged sphere were like currants (dry fruits) in a spherical Christmas pudding. Thomson compared the model of an atom he proposed to a Christmas pudding. This experiment was carried out in the year 1897. An electron was the name given to the negative particle. Thomson discovered the electron, a negatively charged particle, during a cathode ray tube experiment. In 1906, he was awarded the Nobel Prize in Physics for his contribution on the discovery of electrons. Thomson was a British physicist who was born on December 18, 1856, in Cheetham Hill, a Manchester suburb.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
